Very few public health interventions have been as successful as immunizations in providing substantial and highly cost-effective improvements to human health by enabling the control of preventable diseases and death.1,2 Immunization has enabled the eradication of smallpox, lowered the global incidence of polio by more than 99% and neonatal tetanus by 94%.3,4 Since 2020, however, childhood vaccinations have either stagnated or declined globally.5
The quality and safety of vaccines determine the success of immunization to control vaccine-preventable diseases.6 The World Health Organization (WHO) indicates that as much as 25% of all vaccine products reach their destination in a degraded state7 which can lead to the occurrences of vaccine-related adverse events. Failure to deal rapidly and effectively with allegations of these adverse events can undermine confidence in a vaccine and ultimately reduce vaccine uptake.8,9
Even though vaccines are designed to be both safe and effective, Adverse Events Following Immunization (AEFI) can happen and need to be reported in order to take appropriate corrective actions; such as withdrawal of the rotavirus vaccine after confirming its association with intussusception.10 These any untoward medical occurrence following immunization which does not necessarily have a causal relationship to the vaccine, can have a devastating effect on vaccine uptake and increased vaccine-preventable diseases,8,9,11 unless addressed appropriately.
The WHO mandates the systematic collection, analysis, and evaluation of medically important adverse events following immunization.12 Through monitoring and analyzing these adverse events, the health system can detect and respond to safety signals, assess vaccine causalities, and, most importantly, inform public health decision-making.13 In most countries, AEFI detection takes place primarily through routine passive surveillance (spontaneous reporting). This involves vaccine recipients, parents of immunized infants and children, healthcare workers and staff in immunization or healthcare facilities detecting the AEFIs and reporting them to healthcare workers within the system.
Systematic and consistent analysis of vaccine safety surveillance data facilitates a thorough evaluation and dissemination of current information regarding the risks associated with vaccines. This enhances transparency within the immunization program, thereby addressing rumors and mitigating negative perceptions surrounding vaccines. However, in most developing countries, AEFI surveillance and management is limited due to limited awareness, poor pharmacovigilance infrastructure, inadequate skilled human resources, and ineffective coordination, thereby limiting the capacity for effective surveillance.14
Moreover, the introduction of new vaccines, such as those for yellow fever and malaria, in Ethiopia underscores the need for more robust AEFI surveillance systems.15 As these vaccines are administered to the population, robust monitoring mechanisms are essential to promptly identify and address any potential adverse events, ensuring vaccine safety and building public confidence in vaccines.
A comprehensive evaluation of vaccine quality control and safety management practices is essential to mitigate AEFIs and enhance the overall success of the immunization program.7,12 By examining current practices, identifying challenges, and implementing targeted improvements, the AEFI surveillance can be significantly improved thereby enhancing vaccine uptake by the community.7 The aim of this study was to investigate current practices and challenges associated with AEFI surveillance and management in Ethiopia, with the goal of informing evidence-based policies and interventions to improve vaccine safety. The study further provides a detailed analysis of the characteristics of AEFI cases at the community level and the routinely reported AEFIs from health facilities, including the demographic distribution, reporting, types of the AEFI, seriousness and the role of facilities in AEFI surveillance.
METHODS AND MATERIALS
Study design and period
A cross-sectional design was employed to explore the AEFI surveillance system in Ethiopia using both quantitative and qualitative data collection. This study was conducted by a consortium of 5 Ethiopian universities (Addis Ababa, Gondar, Haramaya, Hawassa, and Jimma) in collaboration with the Ministry of Health of Ethiopia. The Immunization Interagency Coordinating Committee (ICC), the National Immunization Technical Advisory Group (NITAG), and the Immunization Technical Working Group served as platforms for stakeholder engagement from inception to validation of the findings. The study was conducted from March to July 2023.
Study Setting
All regions and city administrations of Ethiopia except Tigray (due to the conflict at the time of the study) were included. Ethiopia’s healthcare delivery system is organized into three-tier systems: the first level is the primary healthcare unit (PHCU); comprised of a primary hospital (that serves about 60,000-100,000 population), health centres (serving 25,000-40,000 population) and their satellite health posts (3,000-5,000 population) connected to each other by a referral system. The second level tier is a general hospital covering a population of about 1 to 1.5 million people, and level three is a specialized hospital serving a population of 3.5 to 5 million people.16
An estimated 408 hospitals, 3787 health centres, and 17967 health posts serve as public health institutions in the country. Immunization service is provided at all levels of the health system including at tertiary hospitals, with an estimated 18,000 facilities expected to provide vaccination service. AEFI surveillance is being coordinated by the Ethiopian Food and Drug Authority (EFDA), and health facilities are expected to carry out the activity as part of the immunization service delivery.
Data Collection and Data Sources
Data were collected using different sources, including administrative and health facility assessment, desk review, exit interviews with mothers or caregivers, and interview at households. AEFI experiences were obtained from the exit interviews at health facilities and from household level interview with mothers of children aged 12-35 months (children eligible to receive childhood vaccination), and adolescent girls (for HPV vaccination). Structured interview questionnaires were used to the interview with mothers and adolescent girls individually. The numbers of AEFI reported cases were extracted from the health facilities registers and from interview. Data were collected using ODK uploaded on tablet computers.
The qualitative data that aimed to explore the current practice and challenges associated with AEFI surveillance were obtained from key informant interviews with immunization actors at all levels including the ministry, EFDA, regional health bureaus, woreda health offices, and health facilities. The KII was conducted with vaccinators, and immunization focal persons from health facilities, and the health extension workers. Focus group discussion was conducted with the community members to explore their experience with AEFI. The qualitative study participants were selected purposively using intensity sampling. Maximum variation was used to recruit regions considering Penta 3 or measles vaccination coverage as they are a major indicators of the performance of the immunization program. Data were collected with experts on qualitative study, graduate level education and fluent in local languages.
Sample Size and Sampling Design
A two-stage stratified sampling technique was used to select the study population. In the first stage, enumeration areas (EAs) were randomly selected from stratified (urban and rural) sampling frame prepared for each region by the Ethiopian Statistical Services (ESS) (21.4% urban and 78.6% rural). A total of 471 (101 urban and 370 rural) EAs were studied. All health posts in the selected EAs and their corresponding catchment health centres were also included. Hospitals, WoHOs, and ZHDs serving the enumeration areas were also studied as they are part of the chain of EPI referral links. The ministry’s EPI desk, EFDA, 12 regional health bureaus, 51 zonal health departments, 233 woreda health offices, 71 public hospitals, 314 health centres, and 291 health posts were assessed.
Further, a total of 2,955 exit interviews with health facility attendees and 13,666 household interviews with mothers and caregivers of children were conducted. Additionally, a specific focus on COVID-19 and HPV vaccines was maintained, with 13,874 people aged 12 years or above and 5,245 adolescent girls, respectively, interviewed to assess AEFI experiences related to these particular vaccines.
Measurement and Variables
The number of AEFI cases, types of AEFI, actions taken, the role of health facilities in AEFI surveillance, and compensation for severe AEFI were the major variables measured. In this study, AEFI was defined as the occurrence of any untoward sign that occurred as a result of taking vaccines among children, adolescent girls and individuals 12 years or older. Participants were inquired about the occurrence of any adverse events after receiving vaccinations. Participants were also asked for compensation (in terms of cash, free medical services and for those who experienced severe forms of AEFI. The total numbers of AEFI cases recorded and reported by the health system was extracted from the registers of the health institutions. When there are no documents or registers at the health facilities, it was considered as no AEFI report for that particular facility.
Data Quality Control
Data collection tools were adapted and designed from international and national EPI documents. Data were collected with at least a master’s level education, fluent in local languages and received extensive training for seven days on the objectives, and data collection tools and procedures. Data were collected using the ODK platform, which incorporates built-in quality control measures.
Prior to the main data collection, the study instruments were pre-tested in five kebeles located in five different regions, and involving health institutions separate from the study sites. This pre-test assessed the consistency and validity of the data collection tools, and the resulting inputs were used to refine them. Furthermore, on-site supervisors ensured data consistency and quality before submission to a central server. Daily data uploads were followed by regular feedback based on ongoing preliminary analysis.
Data Analysis
The data were initially cleaned and checked for data consistency. AEFI indicators were calculated as proportions relative to the total number of reviewed health facilities. These proportions and the absolute counts of reported AEFI cases were subsequently aggregated by region, health facility, and setting (urban, agrarian, and pastoral). To provide a comprehensive understanding of the surveillance system, the quantitative findings were supplemented with qualitative data that explored the challenges associated with AEFI surveillance. Regarding the qualitative data, audio-recorded interviews were transcribed verbatim, and notes were taken in the field. A codebook was developed based on the initial review of the transcripts, and transcripts were systematically coded using ATLAS.ti software. Double coding was initially used, with disagreements resolved by discussion; updates to the code definitions were made when needed. Double coding continued until no new disagreements were identified. Thematic analysis described and compared general statements as relationships, themes, and sub-themes emerged in the data. Researchers read all interview transcripts, counterchecked the transcripts, coded the data, and agreed on the emerging themes
Ethical Clearance
Ethical approval was granted by respective Universities’ Institutional Ethical Review Board (IRB/288/15). Prior to data collection, written informed consent was obtained from all participating mothers/caregivers after explaining the study’s purpose, risks, and benefits. In the case of minor participants, written assent was also obtained in addition to parental/guardian consent. Confidentiality and privacy were strictly maintained throughout data collection. The study adhered to the principles of the Helsinki Declaration throughout the study.
RESULTS
Participants’ characteristics
Overall, a total of 26,016 individuals who ever received at least one dose of vaccines were interviewed for occurrence of AEFI. The study included 15,477 children aged 12-35 months who at least received one dose of childhood vaccine, of which 12,524 of them were interviewed at house hold, and 2953 were approached at health facility when they come for vaccination. In addition, 7,658 individual above age of 12 who received COVID-19 vaccine, and 2,881 adolescent girls aged 15-18 years who ever took at least one dose of the HPV vaccine were studied. The largest sample size is contributed by Oromia, followed by Amhara region, the least sample was assigned from Harari, followed by Gambella, Dire Dawa and Benishangul Gumuz (Table 1)
Of the studied children, 70% were from Agrarian areas and about 7% from pastoral areas. Regarding mothers or caregivers of the index child, majority of them (96.5%) were married, 22% were primipara, and 54% were 24 years or younger. Of those interviewed for the COVID-19 vaccine, about 18% were residing in urban areas, 46% had no formal education, 77.7% were between 20-39 years of age, and 92.1% were married. Regarding adolescent girls vaccinated for HPV, 75.4% were rural residents, majority of them (95%) had formal education, and 85% were attending school at the time of the study. Of the interviewees, 68.1% of their mothers and half (50%) of their fathers had no formal education.
Adverse Events Following Immunization
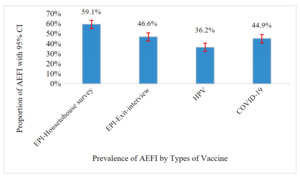
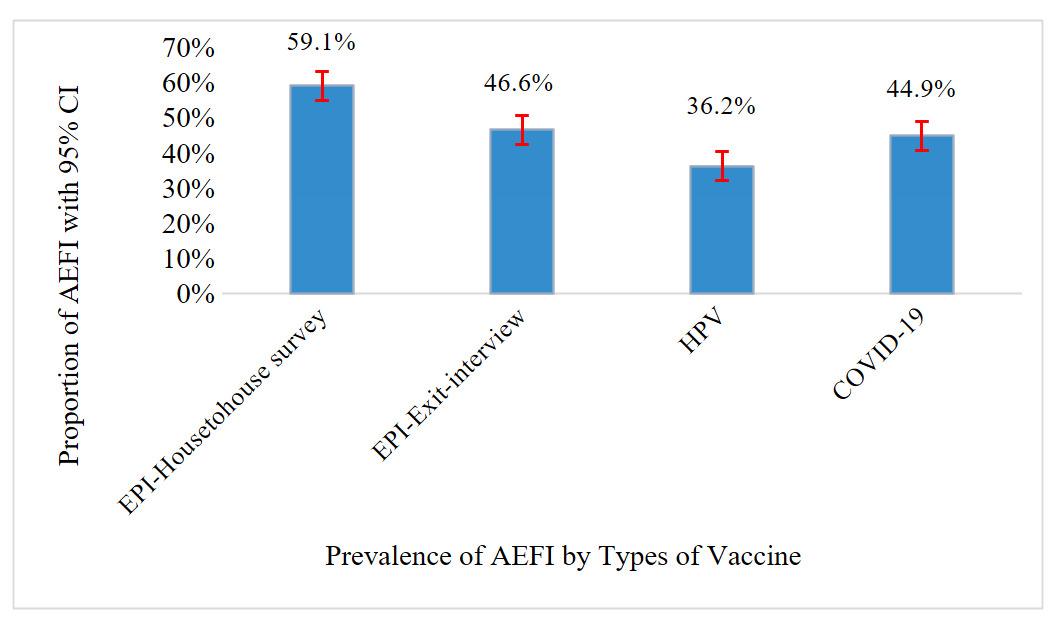
Overall, 8,771 (56.7%) reported at least one adverse event following any of their previous vaccination; 59.1% (95%CI: 58.0-60.1) from household surveys, and 46.6% (95%CI: 44.5-48.9) from exit interviews. Among a total of 7658 individuals who received at least one dose of COVID-19 vaccines, 3,435 (44.9%; 95%CI: 43.6-46.1) developed at least one adverse event. Moreover, of 2913 adolescent girls who received at least one dose of HPV, more than one-third of them 1055 (36.2%; 95%CI: 34.2-38.3) reported to have experienced at least one form of AEFI (figure 1).
AEFI reported from pastoral areas was high for household surveys (60%), exit interviews (57%), COVID-19 (64%) and HPV vaccines (64%). Moreover, AEFI of routine immunization at the community level was reported frequently from Somali (78%), and Afar (73%) regions and less reported from Southwest Ethiopia and Addis Ababa. Similarly, the highest AEFI of COVID-19 was reported from the Somali (83%), followed by Afar region (75%). The highest HPV AEFI was reported in the Somali region (72%), and the lowest from Dire Dawa (26%) (table2).
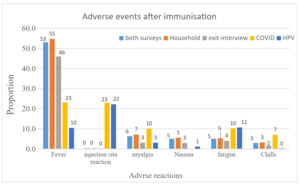
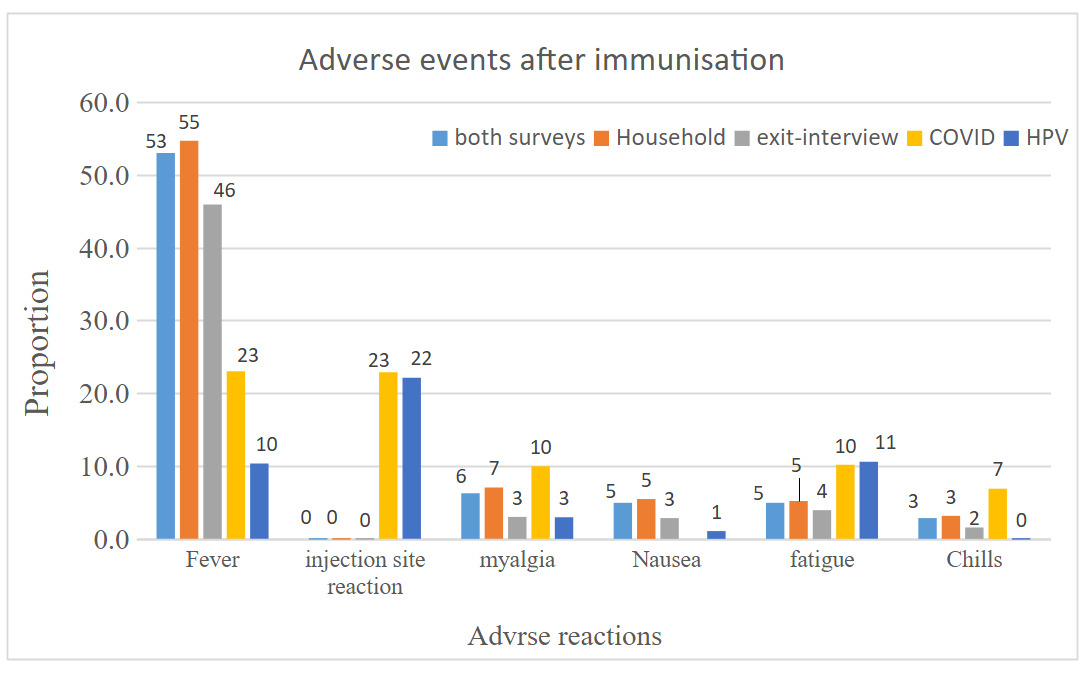
Among children who received routine vaccination, the majority of the symptoms were non-serious adverse events; fever constituted 8,493 (93.5%), followed by myalgia 1,004(11.1 %) and fatigue 793(8.7%). About 12 (0.14%) children were hospitalized, 68 (0.44%) faced life-threatening illnesses, and 9 (0.1%) children were permanently disabled because of AEFI. For COVID-19 vaccine receivers, fever 1760(23%) and injection site reactions 1752(22.9%) were the leading symptoms. The life-threatening complication was reported by 20(0.3) individuals, and thrombosis was reported by 17(0.2%) individuals. For adolescent girls who received HPV, injection site reaction 644 (22.1%), tiredness 308(10.6%) and fever 301(10.4%) are the most reported symptoms. Severe adverse events, including seizures and fainting, are reported among 6(0.2%) and 17 (0.6%) individuals, respectively.
Measures taken for AEFI
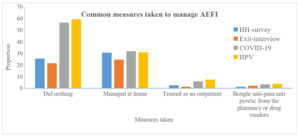
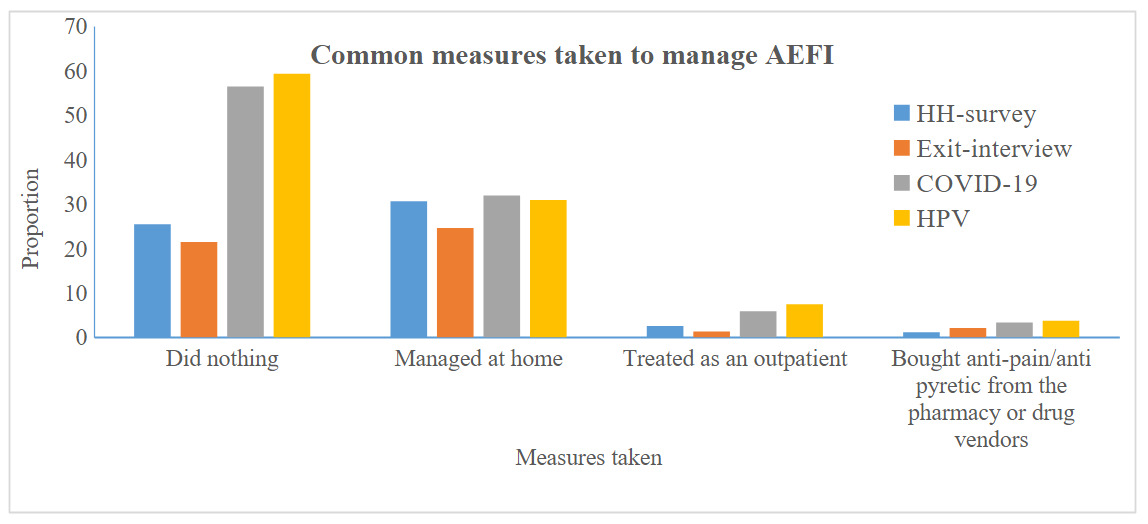
Among the total people who developed AEFI, 61% sought some type of treatment, half (50.6%) managed the cases at home, 6.1% were treated at an outpatient department, 4.4% took anti-pain/antipyretic from a pharmacy, 0.5% sought treatment from religious services. Regarding compensation for the AEFI, 9.1% were compensated with cash, and 11.1% compensated with free treatment. The in-depth interview report further indicated that almost all mild AEFI cases were managed at home, with only few people visiting health facilities, where necessary advice and treatment were provided (figure 3). Finding from qualitative study also showed that individuals have no interest in reporting AEFI, which potentially led to under reporting of the accurate prevalence of AEFI.
“…there was a challenge in reporting of AEFI following COVID-19 vaccination. Most of the vaccinated individuals were not interested in reporting adverse events related to the vaccine despite experiencing them and that results in underreporting of AEFIs from the community” (KII, Health worker)
Compensations in place for AEFI
Those who developed AEFI are expected to benefit from a compensation scheme; however, in this study, few individuals received compensation from their respective bodies. Fifty-eight adolescent girls received compensation for the adverse event following immunization 48 of them received free medical service, and 8 of them received cash compensation. A qualitative report highlighted free medical service provision for AEFI cases. However, the absence of a clear compensation scheme or policy for AEFI cases was one of the barriers to AEFI identification, reporting and surveillance in the sector. Most of the participants mentioned that AEFI compensation policy and mechanism is required for those who suffer from vaccinations, despite the efforts made by health facilities in supporting the cases.
“Compensation is provided when an adverse event is directly linked to the vaccine itself. However, if the issue arises from a health worker’s error, the case is treated as a matter of legal accountability rather than an AEFI. This is happening because of the protocols and implementation of the AEFI compensation scheme remain unclear” (KII, MoH)
AEFI Surveillance and Management Practice at Health Facilities
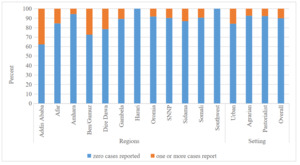
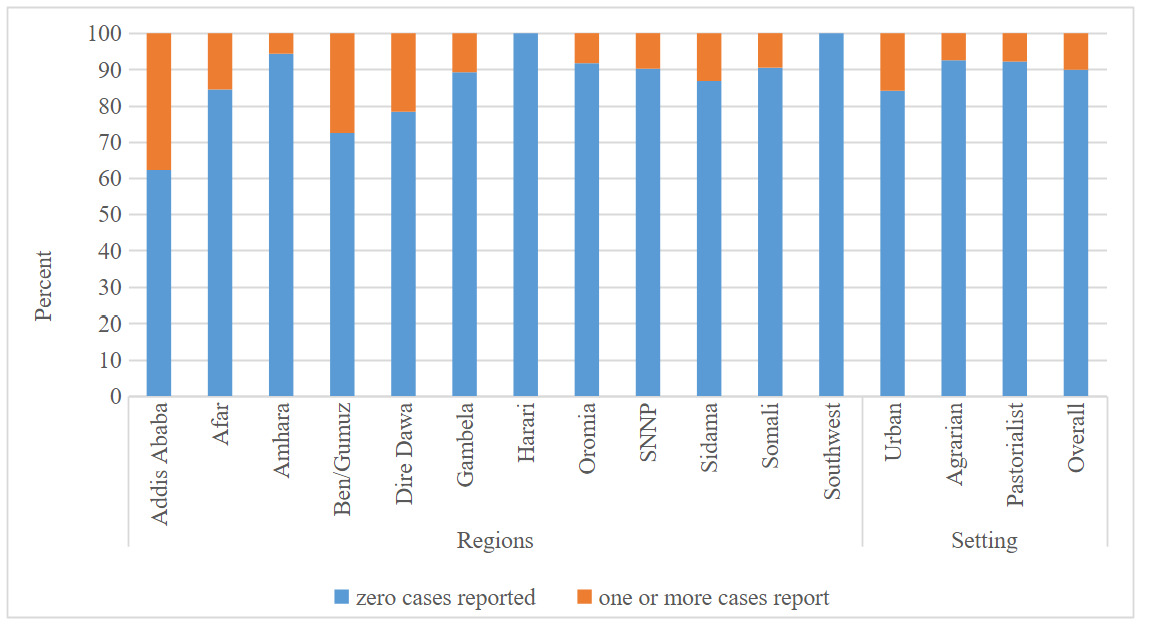
This survey revealed that a significant majority (88%) of facilities have not reported a single AEFI case in the year preceding the survey, with only 4% reporting more than 10 AEFI cases in the same period. While about 40% of facilities in Addis Ababa reported one or more cases of AEFI, Harari and Southwest Ethiopian people regions reported zero AEFI cases one year preceding the survey (Figure 4).
Presence of weak detection and reporting of AEFI at facility and community levels, and health extension workers with limited skills in the detection and reporting of AEFI cases were the major reported challenge from the qualitative study. Failing to conduct regular AEFI case detection in routine immunization and underreporting of AEFI cases among health workers derived from fear of accountability and disrespect from colleagues were also reported.
Health facilities providing immunization services reported the facility’s role in managing AEFI cases. A higher proportion of facilities (64%) had a role in AEFI reporting and notification followed by the prevention and management of AEFI cases. Moreover, 15% of the facilities either have had no role in the management of AEFI or do not know their role. Looking at the disaggregation by the role of facilities by their location, across the board, facilities from pastoral areas had a lower practice of AEFI management and investigation. For instance, less than one-third of facilities from pastoral locations notify and report AEFI cases when they happen, compared to 65% and 68% in urban and agrarian facilities, respectively (Table 3).
Consistent with quantitative findings, the qualitative study revealed that there was a skill gap in the detection and reporting of AEFI at the health posts. It was also pointed out that HEWs at health posts are overburdened with other workloads, and no paid attention to AEFI in the community level. Lack of a focal person for the surveillance activities and high staff turnover made the detection and reporting inconsistent at these facilities. On the other hand, AEFI surveillance activities are not considered part of the routine activities of the health posts.
“We don’t do AEFI surveillance on a regular basis. When we have enough time or less workload, we conduct house-to-house surveillance; to be honest, we don’t consider it as a top priority. However, we advise the community to bring any AEFI cases back to us, including leg weakening and/or flush (red rash) following vaccination” (KII, HEW)
AEFI Surveillance System challenges
This comprehensive evaluation of the AEFI surveillance system revealed several critical shortcomings. Across health facilities, lack of adequate skilled human resources, high turnover of immunization experts and focal persons, health workers skill gap in detection and reporting of AEFI at health posts, fear of reporting and accountability among health workers, workload at health posts, and low motivation of health extension workers were among the major reported challenges. At the federal level, poor coordination among the different actors coordinating AEFI surveillance has been identified to be a critical challenge. The major AEFI surveillance management were categorized as structural (coordination, communication, policy related issues), technical (surveillance data quality, feedback, AEFI detection) and behavioral (knowledge, attitude, skills of health workers) factors as described below.
Structural challenges: Ambiguity in roles and line of communication, absence of AEFI taskforce at zonal and woreda to conduct AEFI surveillance, weak coordination between stakeholders, high workload at health posts, turnover of EPI focal were among the major challenges reported. The AEFI surveillance not being integrated with routine Health Management Information System (HMIS), absence AEFI clear compensation policy, and infrastructure related problems such as electric power and refrigerators were also identified to affect the surveillance system. Although private health facilities provide vaccination in the country, their sub-optimal engagement and deficient reporting of AEFIs further affected the surveillance system. At health posts, workload of the HEW was mentioned as the major reason for not conducting AEFI surveillance. No dedicated person is assigned in these facilities where the majority of vaccinations are provided according to the key informants. Further, vaccine safety concerns were raised as a result of challenges with cold chain of vaccines particularly during outreach vaccination programs.
“…vaccinations provided in outreach sessions takes two or three days to reach the community, at this time, we are worried about the safety and quality of vaccination” (KII, ZHD)
From the demand side, weakening community structures and inadequate engagement of community platforms such as women and health development army in detection and reporting of AEFIs has also negatively affected the program.
Technical challenges: Low reporting of AEFIs during routine vaccinations, data quality problems, absence of regular feedback, non-functional AEFI task force at the regional level, weak detection and reporting at the facility and community, and absence of regular maintenance of faulty refrigerators were identified as technical challenges. A notable limitation identified in the study was the inadequate AEFI surveillance practices during routine immunization. While AEFI monitoring is often prioritized during vaccination campaigns, a more comprehensive approach is necessary to ensure the effective detection and reporting of adverse events throughout the immunization program. Moreover, inadequate counseling of mothers about possible adverse events by health workers was also reported.
Behavioral challenges: Fear of reporting and accountability by health workers, low staff motivation, the skills gap in detection and reporting of AEFI at the health posts, low motivation of HEWs, low awareness in the community, negative perception and rumors, knowledge and skill gap in cold chain management among EPI service providers were among the major behavioral challenges reported (Supportive materials).
DISCUSSION
In Ethiopia, prevalence of adverse event following immunization ranges from 36-59%. More than half (95%CI: 58.04-60.15) children surveyed at household, 46.62% (95%CI: 44.47-48.78) of children interviewed at health facility, 44.86% (95%CI: 43.60-46.11) of those who received COVID-19 vaccines, and 36.23% (95%CI: 34.22-38.30) adolescent girls received at least one dose of HPV reported occurrence of AEFI. A large number of vaccine recipients are concerned about their health related to AEFI.
This findings highlighted a significant yet often overlooked negative impact of adverse events on the immunization programs. People may refuse further immunization, making the individual susceptible to vaccine-preventable disease which is more serious than the AEFI itself.17 Maintaining public trust in vaccine safety is therefore crucial to the success of the program.17,18 Our finding is in alignment with previous studies,19,20 and lower than studies conducted in different countries,21–23 and higher than other studies.16,24,25 The difference in AEFI prevalence among studies is associated with variation in the study site, population type, and size. Some of them are collected from secondary data, others from small population sizes, while others are collected at the national level.
Fever, injection site reaction, and myalgia were the common types of reported AEFI, this common types of AEFI were in congruity with the study conducted elsewhere.20,21,26–28 Moreover, a substantial number of immunization recipients develop a serious life-threatening condition that leads to hospitalization and permanent disability. Unless the AEFI surveillance system is reported timely and managed well, it will lead to public suspicions of vaccines and parents may refuse further immunization for their children.12 Healthcare providers need to be proactive in encouraging them to report AEFI occurrences. If treatment is necessary for a particular condition, the person with an AEFI should be referred to the nearest hospital or health facility.12,17,25,29
Among those who developed AEFI, 61% of them sought some type of treatment from various sources. The majority of them (50.6%) managed the cases at home, 6.1% were treated at an outpatient department, 4.4% took anti-pain/antipyretic from a pharmacy, and 0.5% sought treatment from religious services. Some of the respondents mentioned that they don’t have information on what to do in the occurrence of AEFI. They reported to use the remedies they thought to be good to relieve the symptoms. This finding calls upon vaccination providers at health facilities to provide all important information regarding AEFI, and where to go in case it happens. This finding is supported by other studies conducted in different parts of the world.19–21,27,30,31 Even though compensation and free treatment are considered as one scheme of AEFI protocol, only 9.1% were compensated with cash, and 11.1% are compensated with free treatment for the AEFI.
Against the recommendation of WHO, in this study more than 88% of health facilities have not reported a single AEFI case in the year preceding the survey, and only 4% of them reported more than 10 AEFI cases in the same period. This finding highlighted that serious gap detecting and reporting AEFI by health facilities, which lead to under report the occurrence of AEFI, and miss the opportunity of reporting and managing AEFI. This gap is mostly due to lack of coordination, health workers’ limited knowledge, competing priorities, fear of blame or accountability in case of the AEFI occurs due to error in vaccine handling, error in vaccine prescribing or non-adherence to recommendations for use, and error in administration.25
This finding revealed that AEFI Surveillance system trapped with multifaceted challenge sourced from structure, behaviors and technical capacity of health care providers. The line of communication, mandate and duty is not well assigned among the triparty working on AEFI. Though the activities have been shared among Ethiopian food and drug authority, ministry of health through, and Ethiopian public health institute it lacks coordination and clear communication.17 Structural challenge critically affect management of AEFI. Moreover, technical challenge like lack of assigned human resource, lack of equipment, and role confusion severely affect the process of AEFI management at national level. Furthermore, community’s lack of awareness, health care provider’s skill and knowledge gaps, and lack of motivation directly affect the management of AFI in Ethiopia. This oversight underscores the need for enhanced capacity building and support for healthcare workers to ensure they are equipped to address maternal concerns and provide comprehensive information about vaccine safety. The future success of immunization programs in the country will be hampered unless structural, behavioural and technical challenges are resolved by placing clear mandates and responsibilities among concerned bodies. Other similar studies support our findings.25,31–34
This limitations associated with this study are worth mentioning. Recall bias may affect results from the household interviews, COVID-19 and HPV vaccinations, as participants were asked about their experience with adverse events after they are vaccinated long time before the interview. There might also be documentation gaps that could underestimate reported AEFI cases at health facilities. Additionally, the survey did not include the Tigray region due to the unrest during data period. Despite these limitations, the study provides a nationwide assessment of AEFI surveillance, utilizing a large, diverse sample across various vaccine types and employing mixed methods at both population and facility levels.
CONCLUSION
Substantial number of vaccine recipient in Ethiopia reported occurrence of adverse event following immunization, with majority being non serious conditions. Seeking treatment and managing the case by health facilities showed a critical gap which have prevented clients from taking subsequent dose of vaccine and influence their social network. Significant majority of the health institutions have not reported a single case of adverse events following child, HPV and COVID19 vaccinations. AEFI surveillance system need to be revitalized to address detection, managing, and reporting the case for further intervention to enhance vaccine uptake and improve health of the community in Ethiopia.
ACKNOWLEDGMENTS
The authors would like to acknowledge the Ministry of Health for the support offered throughout the study. Our sincere thanks also goes to regional health bureaus, zonal health departments, woreda health offices, health facilities and study participants across the country for sharing data. We would also like to thank our dedicated field team for their tireless efforts in collecting data across the country. This includes our field supervisors who provided invaluable guidance and support, facility level data collectors who ensured accurate data collection at health facilities, and field data collectors who worked diligently in the communities.
FUNDING
This research was made possible by the financial support of Amref Health Africa. The World Health Organization (WHO), UNICEF, JSI, and PATH, also provided logistical support during data collection.
AVAILABILITY OF DATA
Data are available upon reasonable request.
AUTHORS CONTRIBUTION
All authors contributed to the study. The specific contributions include AA, BH, MD, TG, SS conceptualizing the study, designed the research, data management and analysis, and wrote the manuscript and had primary responsibility for final content. LR, SH contributed to fieldwork, data management & analysis, manuscript writing and critical review of the manuscript. All other authors contributed during design of the study, data collection, and critical review of the manuscript.
COMPETING INTEREST
The authors declare that there is no conflict of interest