Introduction
Worldwide, tuberculosis remains the second deadliest infectious disease after COVID-19.1 It is a bacterial disease caused by Mycobacterium tuberculosis, which most commonly affects the lungs. It is an infectious and contagious disease transmitted directly from person to person, and it carries an enormous social and economic burden for populations.2 Tuberculosis is both curable and preventable. Approximately 85% of individuals who develop the disease can be successfully treated with a 4–6-month therapeutic regimen; treatment also offers the additional advantage of reducing further transmission of the infection.3
In 2021, the World Health Organization (WHO) reported that 48% of people affected by tuberculosis and their households faced catastrophic costs. Since 2015, the same organization has supported numerous national surveys to better understand the burden of costs incurred by patients during tuberculosis care.2 The WHO estimates that 10.6 million people developed the disease in 2021, a 4.5% increase compared with 2020, and that 1.6 million people died from tuberculosis, including 187,000 among individuals living with HIV. The burden of drug-resistant tuberculosis also increased by 3% between 2020 and 2021.1
Many countries across all regions of the world, particularly those in Africa, bear a high burden of the disease. In 2016, 2.5 million people contracted tuberculosis in Africa, and an estimated 417,000 died, representing more than 25% of global tuberculosis-related deaths. The Democratic Republic of the Congo ranks second in Africa in terms of tuberculosis prevalence, with more than 216,000 cases in 2021 and nearly 200,000 tuberculosis cases reported annually.4 The country also faces the challenge of TB–HIV co-infection and is among the four African countries with the highest prevalence of drug-resistant tuberculosis.5
Tuberculosis places a significant economic and social burden, and financial barriers often limit access to healthcare services—both for diagnosis and for completing treatment. In fact, nearly half of patients and their households experience catastrophic costs as a result of the disease.1,6
In 2021, Kinshasa reported 29,990 tuberculosis cases (14% of the national total) and 3,336 TB–HIV co-infections (12%). The PNLT implemented a standardized management protocol in screening and treatment centers (CSDTs), designed to meet population needs.5
The DOTS (Directly Observed Treatment, Short Course) strategy currently applied in tuberculosis care requires patients to attend the health facility daily to take their medication. For treatment to be effective and beneficial to patients, therapy must be completed; otherwise, patients risk developing resistant bacilli and endangering the community. High indirect costs may lead to treatment interruption and, consequently, contribute to bacillary resistance.7
Despite these various findings, the country does not have data on the estimated financial cost of this disease for patients.
Because the financial cost of tuberculosis treatment has not yet been clearly documented, despite its crucial role in ensuring effective disease management; we set out to calculate the actual expenses involved in treating drug-sensitive tuberculosis in Kinshasa, focusing specifically on the Selembao Health Zone.8
Methods
Study design and setting
This was a descriptive cross-sectional study conducted from February 24 to March 17, 2024, at the CSDT of Makala General Referral Hospital (HGR), located in the Selembao Health Zone.
Study population and sampling
The study population consisted of patients with drug-sensitive tuberculosis who were treated at the CSDT of HGR Makala in the Selembao Health Zone between January and December 2023. Inclusion criteria were: diagnosis of drug-sensitive TB at the CSDT Makala; follow-up and classification as cured or treatment completed during the study period; and absence of major associated comorbidities (HIV, diabetes mellitus, COVID-19, etc.). Simple random sampling was applied using the register of patients from the relevant period. Out of a total of 510 recorded patients, 122 were selected using the Random Number Generator (RNG) application.
The sample size was calculated using the following formula:
-
n: Minimum required sample size;
-
Z: 95% confidence coefficient, equal to 1.96;
SD: Estimated standard deviation of the cost in the population. To calculate the SD, we used the mean total cost (USD 527.5) and the range between the minimum (USD 226.3) and the maximum (USD 1,076.7) values of this mean total cost, as reported by Ukwaja et al, Nigeria, published in 2013(8); -
SD = (max − min) / 6 = (1,076.7 − 226.3) / 6 = USD 141.7
-
E: Acceptable margin of error (set at 5%, i.e., 0.05), determined on the basis of the mean total cost, corresponding to USD 26.3.
Thus, n ≥ 111.5.
-
Taking into account a non-response rate of 10%, this corresponds to approximately 11 additional participants, resulting in a final sample size of 122.
To select the study participants, we used the tuberculosis patient register of the CSDT at Makala General Referral Hospital covering the study period from January 1 to December 31, 2023, during which 1,800 patients were recorded. All tuberculosis patients meeting our inclusion criteria were identified and listed in order to create a numbered sampling frame. This process resulted in 510 patients who met the inclusion criteria. From this list, our sample of 122 patients was randomly selected using a random number generator (RNG) application;
Patients who were unreachable or unwilling to participate in the study (n = 3) were replaced by returning to the list of eligible patients and selecting replacements at random.
Study variables
The study included several key variables of interest, summarized as direct costs, indirect costs, total costs, and sources of funding for the management of drug-sensitive tuberculosis. For each main variable, multiple related variables were analyzed.
We adapted the WHO model for estimating economic burden to calculate costs and estimate the total economic burden of managing drug-sensitive tuberculosis, defined as:
Total cost (economic burden) = Direct cost + Indirect cost(6).
Total cost= Direct cost+Indirect cost
- Direct costs:
-
Consultation fees
-
Laboratory tests, imaging, and other fees
-
Medical procedures
-
Nursing procedures
-
Hospitalization fees
-
Medications and laboratory consumables
- Indirect costs:
-
Transportation costs for the patient and caregiver
-
Communication expenses
-
Food expenses for the patient and caregiver
-
Loss of income
Data Collection and Quality Control
For data collection, we used a questionnaire, the majority of whose questions were closed-ended. A guided interview approach was employed, and the questionnaire was explained to the respondents.
The questionnaire comprised five main sections: sociodemographic characteristics, economic characteristics, direct costs borne by the patient, direct costs covered by the National Tuberculosis Control Program (PNLT), indirect costs of care, and sources of financing.
Fieldwork was only possible after obtaining the necessary authorizations through the mission order granted by the School of Public Health of the University of Kinshasa (ESPK). On-site, prior to actual data collection, we obtained approval from the health authorities at the Central Office of the Selembao Health Zone as well as at the Makala General Referral Hospital.
Data were collected using a paper-based questionnaire and subsequently entered into Excel, which allowed for error correction before analysis in the Statistical Package for Social Sciences Version 25 (SPSS v25).
Statistical Analysis
The analyses were primarily descriptive. Quantitative and categorical variables were summarized using tables or charts presenting relative frequencies or percentages. The determination of the different cost categories was performed by calculating minimum and maximum values, as well as the median and interquartile range.
Ethical Considerations
Participation in the study was voluntary. An informed consent form was prepared for this purpose and signed by the respondents prior to the interview. The purpose and importance of the research were clearly explained to the participants. The study protocol was validated by the academic supervisor in 2024 for data collection, while awaiting approval from the Ethics Committee of the School of Public Health, University of Kinshasa (ESPK). Ethical approval was granted in 2025 under reference number ESP/CE/166/2025.
Results
All respondents expected to participate in our study consented to take part in the interview, resulting in a response rate of 100%. The majority of participants (54.1%) were male, with a mean age of 40.7 years; single individuals accounted for 46.7%. Most participants (63.9%) had a secondary level of education, 68% were members of Pentecostal/Revival churches, and traders were the most represented occupational group (27.9%). The majority of respondents (57.4%) had at least three children, and the median monthly income was USD 108, with an interquartile range (IQR) of USD 160 (Table I).
The cost of consultation forms ranged from USD 3.63 to USD 7.27, with an average of USD 3.93 and a standard deviation of USD 0.72. Laboratory examination costs varied widely from 0 to USD 120, with a median of USD 7.45 and an interquartile range (IQR) of USD 3.63. The mean cost of chest X-ray was USD 15.18, with the highest cost recorded at USD 90.91. Costs for other paraclinical examinations reached up to USD 25.45, with an average of USD 3.95.
Similarly, the cost of medications ranged from 0 to USD 218.18, with a median of USD 7.27 and an IQR of USD 24.36. Laboratory tests, radiography, other paraclinical examinations, and non-specific medications had no minimum cost. Costs for RHEZ drugs, RH drugs, laboratory tests, and lymph node aspiration were constant at USD 21.3, USD 20.3, USD 1, and USD 14.55 respectively, due to fixed pricing for these drugs and examinations without variation.
The direct cost of managing tuberculosis varied considerably, ranging from USD 50.6 to USD 286.24, with a median of USD 77.87 (Table II).
It should be noted that communication expenses ranged from USD 0 to USD 20.73, with a median of USD 2.55 and an interquartile range (IQR) of USD 1.45. Most patients spent around USD 1.45 on transportation. Transportation costs for patients ranged from USD 0.36 to USD 19.64, with a median of USD 1.45 and an IQR of USD 1.09. However, for caregivers, transportation costs varied between USD 0 and USD 11.64, with a median of USD 1.45 and an IQR of USD 1.45.
The maximum cost of food rations was USD 9.82 (median USD 1.82 and IQR USD 2.55). Other expenses ranged from USD 0.36 to USD 1.82, with an average of USD 0.85. The loss of income due to a tuberculosis episode ranged from USD 0 to USD 335.4, with a median of USD 167.7. Some patients incurred a minimal indirect cost of USD 2.55 for the entire duration of the illness. For others, indirect expenses could reach up to USD 355.04 for the entire episode.
Half of the patients spent less than USD 174.25 on indirect costs, while the other half spent more. The interquartile range (IQR) of USD 328.58 indicates substantial variability in indirect expenditures (Table III).
Regarding total cost, it ranged from USD 62.6 to USD 607.27, with variations depending on case complexity and the resources required. The median cost was USD 255.39. These costs cover the entire period from hospital admission through diagnosis and treatment, up to completion of therapy (Table IV).
With regard to patient income, 55 participants had an income lower than the median total cost of USD 255.39, while 60 others reported an income higher than the median total cost. This represents a total of 115 out of the 122 respondents. Seven patients reported having no daily or monthly income (Table V).
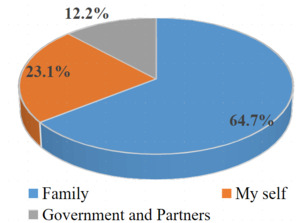
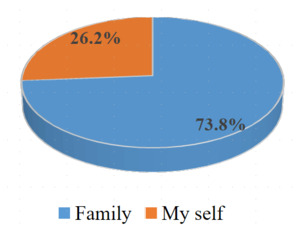
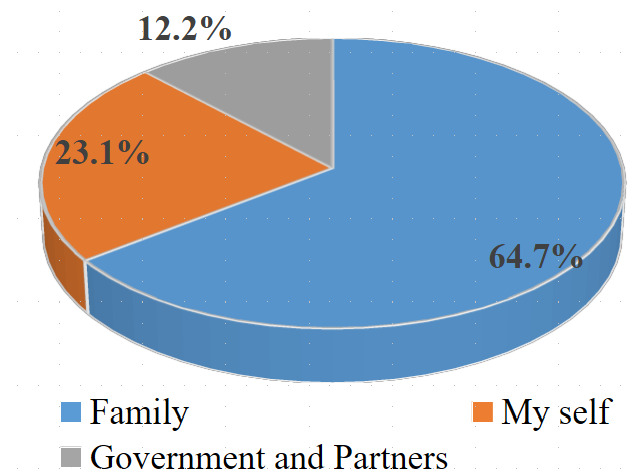
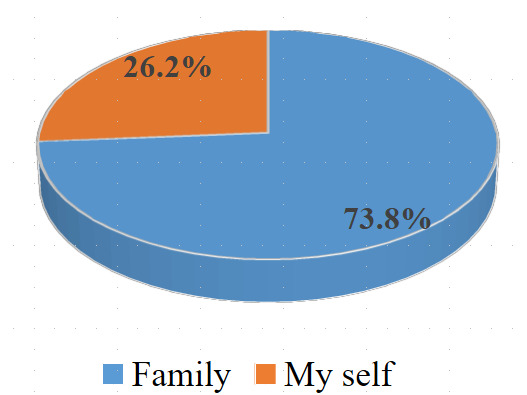
Regarding the sources of financing for patient care, the family constituted the most predominant source of funding despite the subsidies provided by the government and its partners. Aside from the subsidy, for at least 7 out of 10 patients, the family bore the cost of managing drug-sensitive tuberculosis. (Figures 1 and 2)
Discussion
Our study aimed to determine the total cost of managing drug-sensitive tuberculosis in the Selembao Health Zone. According to the findings, the average direct cost of managing drug-sensitive tuberculosis per patient at the Makala Diagnostic and Treatment Center (CSDT), located in the Selembao study area, ranged from USD 50.6 to 286.2, with a median of USD 77.9. The average indirect cost per patient was estimated between USD 2.2 and 2,858.2, with a median of USD 289.5 representing a little more than three-quarters (77.5%) of the total cost of care. The average total cost of care ranged from USD 62.6 to 607.27 with a median of USD 373.3.
Considering the average total cost, our result is far lower than that found in a study conducted in developed countries specifically North America in 2014 on the cost of tuberculosis care, where the cost was estimated at USD 2,000(9). It is also lower compared to another study carried out in Tajikistan in 20079 on the costs borne by households of patients undergoing TB treatment, where the cost reached USD 1,053. These differences may stem from the fact that the first study was conducted in high-income countries, using a meta-analysis that included studies from the USA and Canada. In these countries, input costs reflect a higher standard of living and well-developed health systems. As for the second study, the patients experienced an average two-month diagnostic delay and an average hospital stay of 39 days, during which they received a special diet. These factors could explain the relatively high cost of care. In contrast, in our context, hospitalization is rare, and the diet remains ordinary.
Similarly, our result remains low compared with another study conducted in 2021 on costs incurred by people receiving TB treatment in low- and middle-income countries, where the average total cost per patient for an episode of drug-sensitive TB was estimated at USD 1,253, with a confidence interval of 1,127–1,417 USD.10
According to a study by M. Kaswa et al. on the economic burden of households affected by tuberculosis in the Democratic Republic of Congo, the total cost of managing a first-line TB episode was estimated at USD 400.11 This result, similar to ours, may be explained by the shared context, as the study was also conducted in the DRC, though at a national level.
Our finding is also comparable to a study conducted in 2020 on catastrophic TB costs borne by households in Uganda. According to that study, the mean total cost per patient during a drug-sensitive tuberculosis episode was USD 396 .12 This similarity may reflect the fact that both countries are in Central Africa, sharing common socio-economic and political realities, health system structures, and disease management policies.
A study conducted in Kenya in 2021 on the cost of tuberculosis care services in health facilities reported an average total cost of USD 160 .13 Meanwhile, another study conducted in Egypt between 2019 and 2020 on catastrophic TB costs and their determinants showed that the mean total cost was USD 129.4.14 As seen, these values are lower than those found in our study. This difference may be due to the Kenyan study’s exclusion of additional surveillance test costs (such as chest X-rays and GeneXpert for monitoring disease progression) and other non-specific medications.
Another study conducted in Egypt in 2019 on catastrophic TB-related household expenditures estimated the average total cost of care at USD 198.15 This difference may be explained by the fact that in that study, diagnostic tests, laboratory exams, and medications were covered by a government subsidy, which was not the case in our setting.
Regarding income loss, an WHO survey conducted in 2018 on the cost of tuberculosis to patients estimated the average total cost of care at USD 1,054.16 Income loss accounted for USD 508, or 48% of the total cost. In contrast, income loss observed in our study was USD 167.7, representing 44.9% of the total cost. This difference may be explained by the components used to estimate both direct and indirect costs. Direct costs represent the expenses required to access services, while indirect costs reflect the valuation of human capital, taking into account time spent seeking care and undergoing treatment throughout the TB episode.
According to a study conducted in Nigeria in 2012, income loss per hospitalized patient was estimated at USD 156, while it was USD 114 for non-hospitalized patients. We noted that these values are close to those found in our study, likely due to both studies having been conducted in African settings.13
With regard to subsidies, our study showed that the Congolese government, through the National TB Control Program (PNLT), contributed only 12% of the cost of managing drug-sensitive TB; the remaining portion was covered by partners such as USAID and Cordaid. However, according to a study in Mali, the government of that country contributed 39% to TB management.17
Our results therefore show that the cost borne by patients with tuberculosis is far higher than the contribution of the government and its partners. In the total cost of care, the share of indirect cost which falls entirely on the patient accounts for slightly more than three-quarters of the total, at 77.5%. Given the low GDP per capita in the DRC,18 this total cost remains a significant burden for the Kinshasa population in particular and Congolese households in general.
Although the Congolese government, through the PNLT, declares tuberculosis treatment to be free,17 our findings clearly demonstrate that it is actually a subsidy rather than full coverage. Compared to the burden borne by the government and partners, the family carries 64.7% of the cost of drug-sensitive tuberculosis care.
Strengths and Limitations of the Study
The non-random selection of the Makala CSDT in the Selembao Health Zone means that the findings of our study cannot be generalized. However, given the similar realities across the country, these results despite the limitations may reveal noteworthy trends regarding the subject. Since data were collected retrospectively during interviews, reported expenditures might be approximate, particularly indirect costs, introducing potential recall bias. Considering that the management of drug-sensitive TB lasts at least six months, relying on memory to recall the prices of different services, which is subject to recall bias, could influence certain costs
Conclusion
The findings of this study demonstrate that drug-susceptible tuberculosis continues to impose a substantial financial burden on households, largely driven by indirect costs. Policy responses should therefore go beyond free provision of medical services and include targeted interventions such as transportation vouchers, income-loss compensation mechanisms, decentralization of treatment services and integration of tuberculosis care into broader social protection schemes.
The results of this study will serve as a basis for considering subsidies to support the management of tuberculosis patients.
Ethics statement
Ethical approval for this study was granted by the Ethics Committee of the School of Public Health Approval No. ESP/CE/166/2025. Written informed consent was obtained from all participants prior to their involvement in the research.
Authorship contributions
-
Malonda Ma Ndungi Prince: Conceptualization, Methodology, Formal analysis, Investigation, and Writing original draft.
-
Idiamwana Kutokama Blaise: Software, Formatting, and Writing review and editing.
-
Bijou Masokolo Malamba: Validation, Writing review and editing.
-
Dosithee Ngo-Bebe: Conceptualization, Supervision, and Project administration.
Disclosure of interest
The authors completed the ICMJE Disclosure of Interest Form and disclose no relevant interests.
Correspondence to:
Malonda Ma Ndungi Prince
Department of Planning, Monitoring and Evaluation, Emergency Division, National Program for Emergencies and Humanitarian Action, General Secretariat for Public Health, Hygiene and Social Welfare, Ministry of Public Health, Hygiene and Social Welfare, Kinshasa, Democratic Republic of the Congo
prince_malonda@yahoo.fr