Low birth weight (LBW), defined as a newborn weighing less than 2,500 grams, remains a major global public health concern, contributing substantially to neonatal morbidity and mortality.1,2 Birth weight is a critical indicator of maternal health and nutrition before and during pregnancy, and a strong predictor of infant survival and long-term development.3–6 Globally, UNICEF reported in 2019 that 15% of newborns were LBW, while WHO (2023) noted a nearly stable prevalence of 14.7%, with higher rates in developing countries (16.5%) compared to developed ones (7%).2,3,5,7 LBW infants face a mortality risk about 20 times greater than normal-weight infants.3 In the medium and long term, LBW is linked to cognitive and physical deficits, as well as chronic diseases such as hypertension, diabetes, and nutritional disorders.3,8–12
Sub-Saharan Africa records the highest LBW prevalence, with live births increasing from 4.4 million to 5 million due to demographic dynamics.12,13 In the Democratic Republic of Congo (DRC), prevalence ranges between 6.3% and 13.6%, largely attributable to prematurity and intrauterine growth restriction (IUGR).11,12 National surveys reported rates of 7.1%4 and 10%,14 while local studies found higher figures, such as 14.5% in Mbuji-Mayi and 15% in Lubumbashi.12,13,15 In Kinshasa, prevalence was 7.6% in 2023.16 The Sustainable Development Goals (SDGs) target a 30% reduction in LBW prevalence by 2025.17
LBW reflects both prematurity and IUGR, each with distinct determinants.18,19 It is associated with complications including respiratory distress, infections, and developmental delays.18,20 Contributing factors include socio-economic disadvantage, poor maternal nutrition, and illnesses such as hypertension and anemia.11,21 These conditions expose newborns to hypothermia, perinatal asphyxia, infections, and impaired cognitive development.15,22
Despite extensive documentation of LBW globally and nationally, evidence remains limited on how this issue manifests within specific health systems in Kinshasa. The Salvation Army Medical Service plays a unique role in caring for vulnerable populations, often serving communities with restricted access to other facilities. As a contracted service of a church working in partnership with NGOs, it provides a strategic setting for research. Understanding LBW prevalence and associated factors in these maternity wards is crucial to inform targeted interventions and evaluate whether these structures adequately meet population needs.
This study aims to identify factors associated with LBW in maternity wards of health facilities under the Salvation Army Medical Service in Kinshasa
II. METHODS
STUDY TYPE, PERIOD, AND SETTING
An analytical cross-sectional study was conducted from April 3 to June 29, 2025, within the network of the Salvation Army Medical Service in Kinshasa. Seven health facilities with maternity services were included: BOYAMBI Hospital, Major LEKA Hospital, ESENGO Health Center, BOMOI Maternity, BAKIDI Health Center, NGOLU Health Center, and AMBA Health Center. The study duration of nearly three months was determined by the number of investigators available and the time required to reach the planned sample size.
STUDY POPULATION
The study population consisted of women who delivered in the maternity wards of the selected Salvation Army facilities in Kinshasa.
INCLUSION AND EXCLUSION CRITERIA
Eligible participants were women aged 15–49 years who consented to participate and delivered in one of the study facilities. Women unable to respond to the questionnaire were excluded.
SAMPLING
The sample size was calculated using the single population proportion formula, with an estimated LBW prevalence of 16%, a 95% confidence interval, a 5% margin of error, and a 10% non-response rate. This yielded a minimum of 227 participants. To account for differences in patient volume across facilities, the sample was weighted and expanded proportionally, resulting in a final sample size of 603 women across the seven sites.
VARIABLES AND OPERATIONAL DEFINITIONS
The dependent variable in this study was low birth weight (LBW), defined as less than 2,500 grams.1,2 Independent variables encompassed sociodemographic factors such as maternal age (<20, 20–34, 35–44, ≥45 years), education, occupation, marital status, religion, household daily expenditure (<4,500; 4,500–10,000; >10,000 CDF), and socioeconomic position (poor, middle, rich). Obstetric characteristics included parity (<5 or ≥5 pregnancies), hemoglobin level at first antenatal care (<9 or ≥9 g/dL), antenatal follow-up (yes/no), delivery mode (vaginal, cesarean, instrumental), child’s sex (male/female), and neonatal outcome within seven days (alive/death).
Clinical variables comprised maternal history and conditions such as previous LBW child, hypertension, diabetes, anemia before pregnancy, HIV infection, malaria during pregnancy, pre-eclampsia, and urinary tract infection, all recorded as present or absent. Lifestyle factors included tobacco and alcohol use during pregnancy, while maternal nutritional status was assessed using body mass index (BMI <19, 19–24, 25–29, ≥29). These operational definitions provided a structured framework for analyzing determinants associated with LBW.
DATA COLLECTION
Data were collected using a standardized electronic questionnaire programmed in KoboCollect on smartphones and tablets. The tool captured site identification, maternal sociodemographic and clinical characteristics, and newborn outcomes. Direct interviews were conducted with mothers over 90 days. Investigators received two days of training, supervised by two field coordinators under the principal investigator. A pretest was conducted in two non-study facilities, and adjustments were made. Daily quality checks ensured completeness, accuracy, and consistency.
DATA PROCESSING AND ANALYSIS
Data were stored on a secure server, cleaned in Excel, and analyzed using SPSS version 27. Categorical variables were summarized as frequencies and percentages. Continuous variables were presented as means with standard deviations if normally distributed, or medians with interquartile ranges otherwise. Associations with LBW were first tested using chi-square. Variables with p <0.20 were included in bivariate analyses and subsequently entered into multivariate logistic regression to identify independent predictors of LBW. Binary logistic regression was applied with 95% confidence intervals. Collinearity was reassessed, particularly between prematurity and intrauterine growth restriction (IUGR). Certain analyses were repeated to refine results and strengthen validity.
ETHICAL CONSIDERATIONS
The study protocol was approved by the Ethics Committee of the School of Public Health, University of Kinshasa (Approval No. ESP/CE/59/2025). Participation was voluntary, based on informed verbal consent. Ethical principles of respect for persons, beneficence, and justice were upheld, ensuring equal opportunity for all eligible women. Confidentiality was maintained through anonymization of data.
III. RESULTS
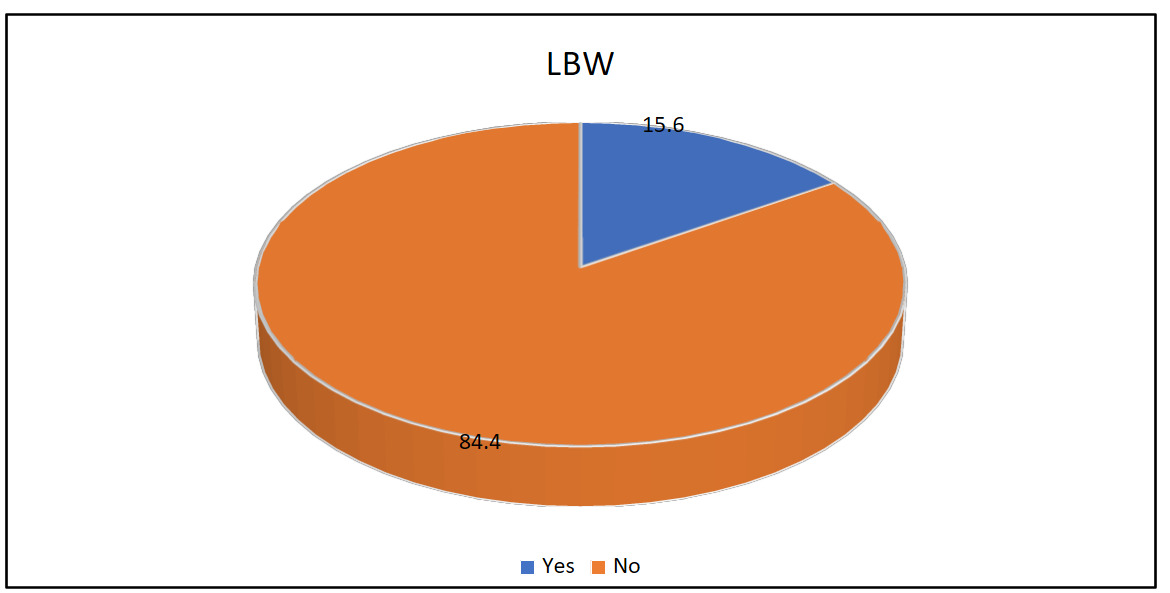
Among 603 women delivering in Salvation Army facilities in Kinshasa, 15.6% had low birth weight (LBW) infants. Sociodemographic factors such as age, education, and occupation showed no significant associations with LBW. Strong predictors included history of LBW, HIV infection, preeclampsia, malaria, and urinary tract infections. Hypertension before pregnancy also increased risk, though it lost significance after adjustment. Neonatal deaths within seven days were more frequent among LBW infants, underscoring their vulnerability.
Among the variables analyzed, several showed statistically significant associations with low birth weight (p < 0.05). Women with a previous history of delivering a low birth weight infant were far more likely to have another LBW baby in the current pregnancy (p=0.001). Likewise, maternal hypertension before pregnancy was significantly associated with LBW (p=0.004). Infections also played a role: mothers with HIV infection (p=0.001), those who suffered from malaria during pregnancy (p=0.01), pre-eclampsia (p=0.001), and urinary tract infections (p=0.005) all had higher proportions of LBW infants. Finally, the outcome within seven days after birth was significant, with neonatal death being more frequent among LBW infants (p=0.002). These findings highlight that both maternal medical history and pregnancy complications are strongly linked to the occurrence of low birth weight in this population.
In both bivariate and multivariate analyses, several determinants remained significantly associated with low birth weight. A history of LBW showed the strongest effect, with mothers nearly 12 times more likely (ORa=12.15, p<0.001) to deliver another LBW infant. HIV infection was also a robust predictor, increasing the odds about fourfold (ORa=4.20, p=0.001). Similarly, pre-eclampsia raised the risk almost fourfold (ORa=3.85, p=0.002). Urinary tract infection doubled the risk (ORa=2.10, p=0.015), while malaria during pregnancy remained significant (ORa=1.65, p=0.042). Other factors such as hypertension before pregnancy (ORa=1.95, p=0.065) and poor socioeconomic status (ORa=1.85, p=0.180) lost statistical significance after adjustment. These findings highlight that maternal medical conditions and pregnancy complications are key independent contributors to LBW in this population.
IV. DISCUSSION
The key findings of this study indicate that the prevalence of low birth weight (LBW) was 15.6%, with several maternal and clinical factors significantly associated with LBW. In both bivariate and multivariate analyses, a history of LBW, HIV infection, pre-eclampsia, malaria during pregnancy, and urinary tract infection emerged as independent predictors, while hypertension before pregnancy and neonatal outcome within seven days showed borderline associations.
PREVALENCE OF LBW
The prevalence of LBW in our study (15.6%) is consistent with results reported by Kangulu IB in Kamina, DRC (14.3%)23 and Ilunga PM in Lubumbashi, DRC (15%).24 This similarity may reflect comparable maternal practices and health conditions across these settings. Our prevalence was higher than that reported by the MICS-DRC 2017–2018 survey (7.1%)4 and by Lufungula Bwaya (9.5%).25 These differences may be explained by methodological variations: the MICS survey relied on retrospective documentation, which may underestimate LBW, whereas our study collected data directly at delivery. Conversely, our prevalence was lower than that found by Nsamput KJ in Kisangani (39%)26 and in Maniema Province (27%),27 likely reflecting differences in socioeconomic conditions and maternal health profiles.
Beyond simple comparisons, variability in LBW prevalence across studies may stem from methodological issues such as sampling strategies, measurement errors, and classification bias. Hospital-based studies, like ours, may overrepresent complicated pregnancies, while national surveys may underreport LBW due to reliance on records. Confounding factors such as maternal age, parity, and socioeconomic status also contribute to differences. Thus, prevalence estimates must be interpreted within the context of study design and population characteristics.
DETERMINANTS OF LBW
Our study revealed that maternal undernutrition during pregnancy increased the likelihood of LBW nearly eightfold. This finding is consistent with Kakudji Prosper et al. in Lubumbashi24 and Telly N.O in Mali,28 who also reported strong associations between poor maternal nutrition and LBW. Undernutrition restricts fetal growth by limiting nutrient supply, but this relationship should be interpreted cautiously. Nutritional status is difficult to measure precisely and may be subject to misclassification. Reverse causality is also possible, as women carrying growth-restricted fetuses may appear undernourished at delivery. Infections and socioeconomic conditions may further confound this association.
A history of LBW was another strong predictor, with affected women being 7.4 times more likely to deliver another LBW infant. This aligns with findings from Maty DC et al. in Senegal,29 who reported a sixfold increased risk. This non-modifiable factor likely reflects persistent maternal health problems that are inadequately addressed during pregnancy. Its recurrence across contexts underscores the need for closer monitoring and targeted interventions for women with prior LBW deliveries, as they represent a high-risk group.
Prematurity also emerged as a major determinant, with preterm infants being 15 times more likely to present LBW compared to term infants. This result is consistent with Saasita K.A in Kisangani, DRC [56], who found a 5.5-fold increased risk among infants born before 37 weeks. LBW arises from either prematurity, intrauterine growth restriction (IUGR), or both. Premature infants are particularly vulnerable because birth occurs during a period of rapid intrauterine growth, and shortened gestation limits fetal development.30–32
Other conditions such as malaria and pre-eclampsia, although not statistically significant in multivariate analysis, remain highly prevalent and clinically important. Their lack of significance may be due to overlapping pathways with other risk factors or limited statistical power, but they should not be dismissed. Similarly, the absence of association with antenatal care attendance, maternal education, and household income warrants reflection. It may suggest that access alone is insufficient if the quality of care is poor, or that socioeconomic indicators do not fully capture maternal vulnerability. These findings highlight the multifactorial nature of LBW, where biological, social, and health system factors interact
STRENGTHS AND LIMITATIONS
This study identified key factors associated with low birth weight (LBW), highlighting modifiable aspects such as maternal undernutrition, which are useful for preventive interventions. A notable strength is the relatively large sample size of 603 women, providing robust data and contributing to the limited evidence on LBW in Kinshasa. However, limitations include the non-random, hospital-based sample that restricts generalizability, short data collection period, and potential recall or measurement bias. The cross-sectional design also precludes causal inference, and modeling prematurity complicates interpretation.
CONCLUSION
This study found a 15.6% prevalence of low birth weight (LBW) in Kinshasa, with maternal history of LBW, undernutrition, and preterm birth as key determinants. Actionable priorities include strengthening nutritional support during antenatal care, closer monitoring of women with prior LBW deliveries, and reinforcing protocols to prevent prematurity. Within the Salvation Army Medical Service, targeted nutrition, risk-based follow-up, and improved clinical management could reduce LBW and improve neonatal outcomes.
Acknowledgements
We are grateful to all the women who delivered in the maternity wards of the Salvation Army Medical Service in Kinshasa and their partners for their voluntary participation. We also thank the investigators and healthcare providers for their assistance and cooperation.
Disclaimer
No disclaimers to declare.
Ethics Statement
This study was approved by the Institutional Ethics Committee of the School of Public Health, University of Kinshasa ((Approval No. ESP/CE/59/2025), informed consent was obtained from all participants involved in the study.
Data Availabilityn
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This research received no external funding. The article publication charge (APC) was not funded by any external source.
Authorship Contributions
-
Serge Musasa: Principal investigator; conceptualization, study design, and overall coordination.
-
Bernard-Kennedy Nkongolo: Drafting of manuscript, statistical analysis, and interpretation of results.
-
Daddy Bopila: Data collection and contribution to manuscript preparation.
-
Peguy Mukieme: Editing of manuscript, support in data analysis, and validation of findings.
-
Nelly Dikamba: Senior investigator; supervision of all study processes, critical review, and final approval of the manuscript.
Disclosure of Interest
The authors completed the ICMJE Disclosure of Interest Form (available upon request from the corresponding author) and disclose no relevant interests