BACKGROUND
Pneumonia is a leading cause of morbidity and mortality in children under five years of age, with an estimated 138 million pneumonia episodes occurring every year. The estimated annual incidence of clinical pneumonia in developing countries was 231 episodes per 1,000 child-years of observation in 2015, which declined from 329 episodes in 2000.1 In 2015, 16.4 million children were admitted to hospitals due to pneumonia, representing a substantial rise from 5.7 million in 2000.1 In 2021, pneumonia caused over 725,000 deaths in children under five years of age.2 Sub-Saharan Africa and South-East Asia, where coverage of basic health services is low, often face inadequate service availability and readiness of health facilities, and sub-optimal quality of care, contributing to more than three-fourths of pneumonia-related deaths. Ethiopia was one of the five countries that accounted for about 50% of the global childhood pneumonia-related mortality.3,4
The World Health Organization (WHO) has three levels of guidance for managing childhood pneumonia: district hospital, primary care health facility, and community level.5,6 At the community level, where most pneumonia deaths occur, WHO and UNICEF have developed the integrated community case management (iCCM) strategy for Community-Level Health Workers (CLHWs).6,7 The WHO guidelines recommend that children 2-59 months of age with chest indrawing or fast breathing and young infants 7-59 days of age with only fast breathing be treated with oral amoxicillin. In contrast, children with any danger sign should be referred to a referral facility.8,9 However, a high rate of non-compliance with referral advice has been reported in studies from low- and middle-income countries (LMICs).10,11
Prompt recognition of pneumonia symptoms and timely care-seeking are essential for reducing pneumonia-related mortality among children under five years of age, which is a key component of the WHO/UNICEF Global Action Plan for the Prevention and Control of Pneumonia and Diarrhoea (GAPPD) framework.12 However, several factors, such as caregivers’ limited knowledge of symptoms and danger signs, reliance on traditional remedies, long distances to health facilities and the cost of care, can hinder their ability to seek appropriate and timely treatment.13,14 Care-seeking rates are inversely related to the distance to the closest health facility. Inappropriate care-seeking, including traditional care, is also common.15,16 The role of CLHWs is crucial in improving care-seeking behaviours, providing prompt treatment, and thereby reducing pneumonia mortality in children, especially in low-resource settings where access to a health facility is limited.17–19 Reduction in mortality and increased care-seeking and treatment for pneumonia, malaria, and diarrhoea through CLHWs was recently demonstrated by the WHO Rapid Access Expansion iCCM programme in the Democratic Republic of Congo, Malawi, Niger, and Nigeria.10,20 Two multi-country trials (Ethiopia, Bangladesh, India, Malawi) demonstrated that CLHWs effectively and safely managed pneumonia with oral amoxicillin in young infants 7-59 days old with fast breathing and in children 2-59 months old with chest indrawing.13,14 Ethiopia has a supportive policy for implementing community case management of pneumonia. However, the scale of implementation is very low, with <1% coverage reported nationwide.21–23
This implementation research will take place in the Tigray region, Ethiopia. Tigray is transitioning out of a two-year conflict that has severely impacted the region and ushered in a challenging period. Affected healthcare facilities reported that medical supplies were severely low and often impossible to replenish, making it difficult to carry out even the simplest healthcare interventions. Progress in restoring healthcare services has been slow, with the extent of damaged infrastructure, looted resources, and a depleted healthcare workforce being significant and challenging to recover from. Many health facilities, particularly in rural regions, continue to face difficulties providing basic services because primary healthcare, the backbone of the system, was severely affected during the conflict.24,25 It further underscores the urgent need to assess the lessons learned and strategies required to implement pneumonia management in a post-conflict setting.
This implementation research aims to evaluate the barriers and enablers for community-based pneumonia management, facilitate policy dialogue, and develop a scalable implementation model. The model will be developed through an iterative cycle of development, implementation, and evaluation, utilising a quality improvement approach to ensure high coverage and effectiveness. The study also intends to develop and implement a scalable model to increase the coverage of pneumonia treatment among children under five to 80% in the Tigray Region. Additionally, it will identify facilitators and barriers to treatment across all levels of the health system, including referral hospitals, general hospitals, and primary health care units [primary hospitals, health centres, and health posts/Health Extension Workers (HEWs)]. The study will also determine the rate of clinical treatment failure and adherence to pneumonia treatment among 7-59-day-old young infants with fast breathing and 2–59 months old children with fast breathing and/or chest indrawing pneumonia treated by HEWs. Additionally, it will explore the knowledge, perceptions, and experiences of health facility staff, HEWs, women development group leaders, and beneficiaries regarding the implementation of pneumonia management. The implementation will be designed for a post-conflict setting in which the health system has been severely damaged by the war. Thus, this study will be crucial in identifying potential barriers and developing effective implementation strategies to scale up the community-based management of pneumonia in post-conflict contexts.
METHODS
Study site
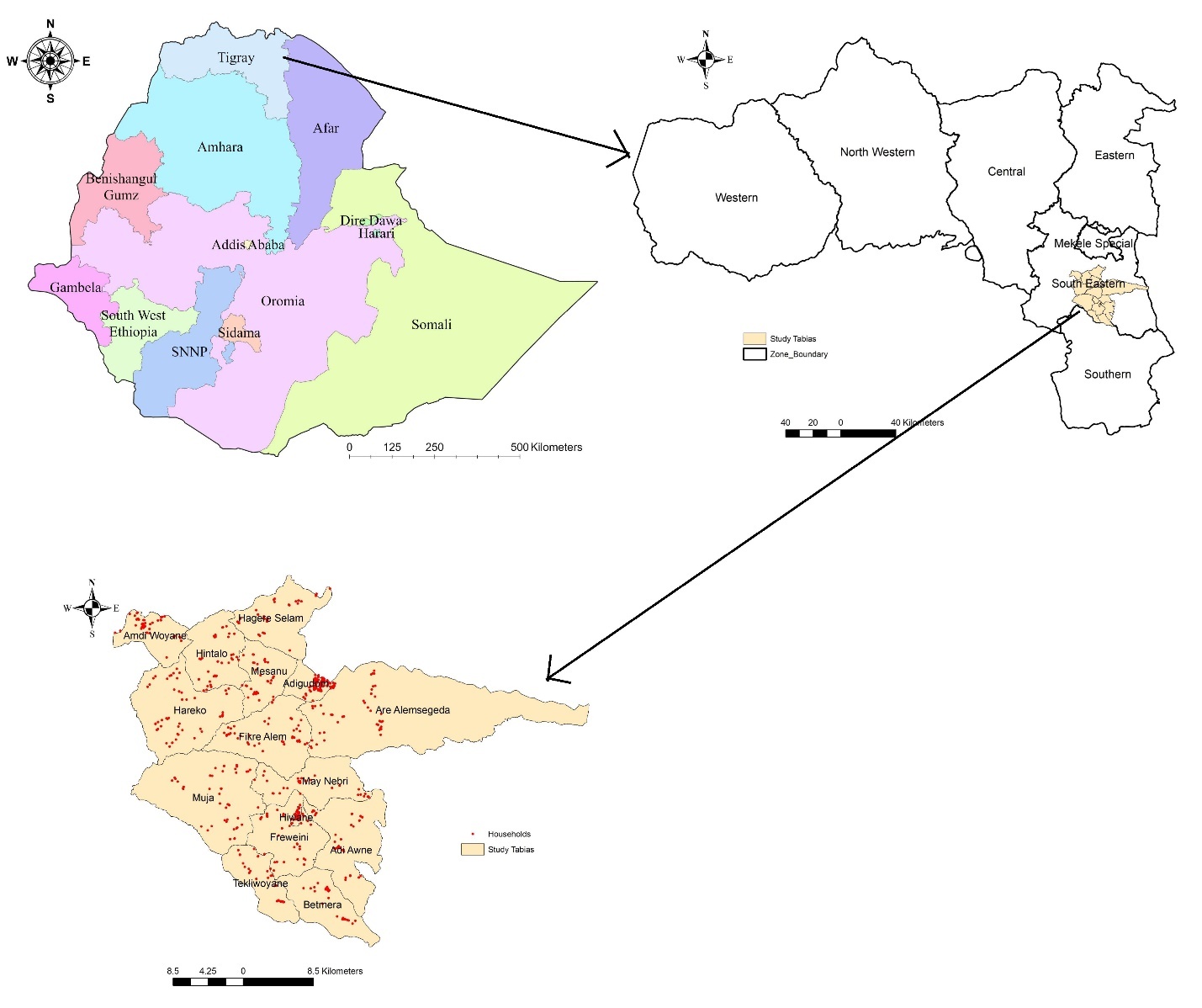
The implementation research will be part of a multi-country study that includes Malawi, Bangladesh, India, and Ethiopia. Tigray is the northernmost region of Ethiopia with an estimated total population of 5,936,000 according to the Ethiopian Central Statistics Agency’s 2024 projection. Rural areas remain home to 80% of the people.26 The Hintalo and Adigudom districts in Tigray, Ethiopia, will be the sites for this study. These districts were selected based on a high pneumonia caseload reported by the Tigray Regional Health Bureau(RHB). There is one primary hospital, six health centres, and 17 health posts in the Hintalo and Adigudom districts. The total population of both districts is estimated to be about 189,713.26 This implementation research study will include all primary health facilities in the district (one primary hospital, four health centres, and 17 health posts).
Study Design
We will employ a mixed-methods study combining qualitative and quantitative methods to evaluate delivery strategies to increase access to pneumonia treatment among children under five years of age.
The implementation research was initially intended to begin in January 2021; however, the conflict in Tigray resulted in unavoidable delays. Ethical clearance was renewed on May 2, 2023, and the project documentation was resubmitted to Mekelle University on May 14, 2023. The official commencement date of the project was May 15, 2023.
Population
The study population will be children under five years of age with pneumonia in the study catchment area. Young infants 7-59 days old with fast breathing (respiratory rate 60 breaths or more per minute) and 2-59 months old children with fast breathing (respiratory rate 50 breaths or more per minute for 2-11 months and 40 breaths or more per minute for 12-59 months old) and/or chest indrawing will be included. Young infants 0-59 days olds with very severe disease/possible serious bacterial infections [not able to feed at all or stopped feeding well, convulsions or fits, movement only when stimulated or no movement at all, severe chest indrawing, high body temperature (≥38°C), low body temperature (<35.5°C) or very low weight for age (< 2000 grams), local infection (red or draining umbilicus, skin pustules), yellow soles (jaundice) or hypoxaemia (oxygen saturation (SpO2) <90% using a pulse oximeter)], or young infants <7 days old with fast breathing will be referred to a higher facility and will not be treated by HEWs. In 2-59 months old children, those with severe pneumonia or very severe disease [danger signs: convulsions or fits, unable to drink or feed, persistent vomiting, unusually sleepy or unconscious, stridor in a calm child, fever (temperature 38°C) or above for 7 days or more and hypoxaemia, severe dehydration, persistent diarrhoea, severe malnutrition, or signs of severe infection] will be also referred to a higher facility.
Intervention
The intervention will consist of identification, assessment, and treatment/referral of children under five years of age with pneumonia at primary health care units (health posts, health centres, and primary hospitals). HEWs will assess and classify a child either in the community or at the health post according to the National guidelines for pneumonia classification.27 Currently, the HEWs identify and treat 2–59-month-old children with fast breathing with oral amoxicillin. The HEWs will use enhanced Integrated Community Management of Newborn and Childhood Illnesses (iCMNCI) (with the additional use of a pulse oximeter) and treat young infants 7-59 days old with fast-breathing pneumonia with oral amoxicillin for seven days and children 2-59 months old with chest indrawing pneumonia for five days with oral amoxicillin (Table 1). A child with any danger signs will be referred to a higher health facility. The proposed frameworks and implementation models will be refined and validated through formative assessment and policy dialogues with the regional health bureau. Formative research will be conducted to identify potential barriers and enablers to implementing the proposed model. Subsequently, implementation and process evaluation will be performed, and the initial model will be refined through continuous learning at each stage until a final, scalable model is reached. The final model will be validated and approved through a policy dialogue with the Ministry of Health and the Tigray Health Bureau.
Conceptual Framework
At the core of our intervention is the conceptual framework outlined in Figures 2 and 3, which emphasises the role of HEWs in identifying and managing pneumonia at the community level. HEWs are trained and equipped to recognise signs of pneumonia, such as cough, fast or difficult breathing, and other danger signs, either during home visits or outreach clinics. Based on their initial assessment, children will be treated either with oral amoxicillin or referred to health facilities if they have any danger signs. If referral is not feasible, young infants will be treated according to the WHO guidelines for possible serious bacterial infection (PSBI) for outpatient management.9,28 This framework ensures that health workers are actively engaged in early identification and treatment, which is critical to reducing pneumonia-related morbidity and mortality.
Implementation Approach
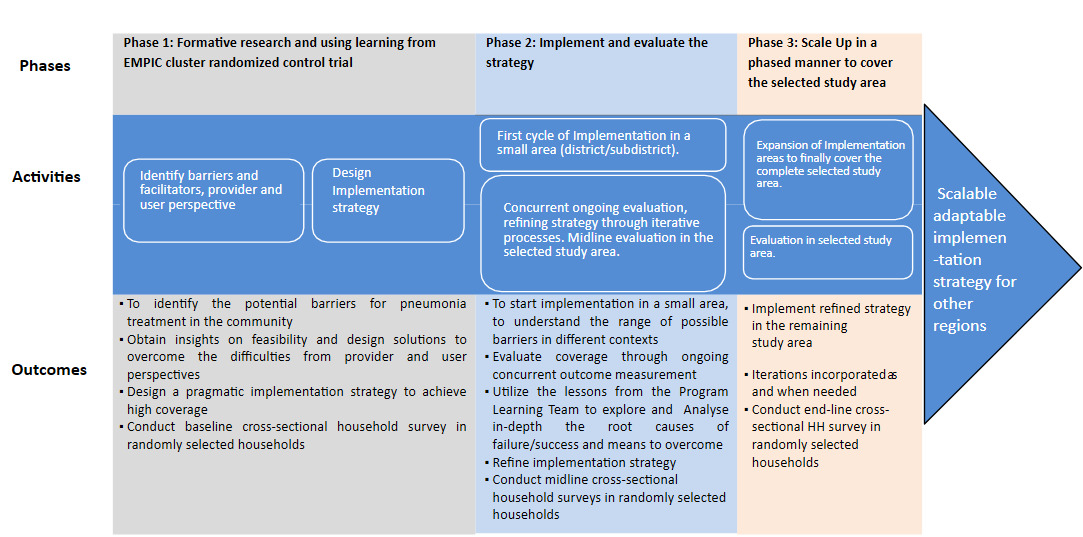
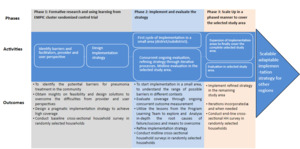
The logical model for this intervention outlines a structured path from initial research to large-scale implementation. The model is divided into three phases: formative research, implementation and evaluation, and scaling-up, each critical for developing, testing, and refining the intervention.
As shown in Figure 4, the model employs an adaptive approach, using real-time feedback to refine the intervention and ensure it aligns with community needs.
Logistical and Infrastructural Considerations
Successful implementation of the enhanced pneumonia management protocol requires attention to key logistical and infrastructural factors. These include consistent availability of oral amoxicillin at health posts, functional and reliable pulse oximeters with paediatric probes, timely referral systems despite transportation challenges in post-conflict districts, and adequate supervision and reporting structures for HEWs. Facility readiness and workforce capacity will also be considered.
Study teams
Overall, the enhanced pneumonia management intervention will be delivered by healthcare workers who are health system employees. The research team’s implementation role remains supportive. The research team will be composed of three teams: i) the programme learning team (PLT), that will conduct formative research and process monitoring that would help formulate the intervention and continuous process evaluation of implementation; ii) the implementation support team (IST), it will be led together with the regional health bureau and will guide the health system to support the implementation; and iii) the outcome measurement team (OMT) will measure the study outcomes independently.
The implementation research will be jointly led by clinical and scientific principal investigators supported by multidisciplinary teams. The three coordinated team structures will be as follows:
Programme Learning Team (PLT): Led by senior public health researchers, will conduct formative research, document implementation processes, and generate continuous learning to refine strategies.
Implementation Support Team (IST): Co-led with the Regional Health Bureau (RHB), will ensure clinical mentorship, supportive supervision, and strengthening of service delivery structures across health posts and primary facilities.
Outcome Measurement Team (OMT): Independently evaluate the outcomes and fidelity of implementation, including quality of assessment and treatment for pneumonia at all levels.
The teams will include medical doctors, paediatricians, public health experts, and programme managers. All researchers receive ongoing technical and mentorship support from the WHO senior researchers, programme experts and paediatricians.
The study will be implemented in three phases, as shown in the logic model (Figure 4). The first phase will comprise formative research to identify barriers and facilitators to the implementation of pneumonia treatment at the community level (iCMNCI) and at the primary level healthcare facility (IMNCI). The formative research will help us identify gaps that need strengthening, which we will do jointly with the RHB. The second phase will involve implementing and evaluating the strategy. It will be used to explore and analyse in depth the root causes of failure/success and the means to overcome them. Based on this, we will refine the implementation strategy. The third phase will involve scaling up in phases to cover the entire study area.
The expected activities will be organised into three alignments. Pre-facility will focus on the community education and case finding of pneumonia (Table 3). As for the facility stage, healthcare workers will be trained and supervision will be conducted at the district level. This stage will involve enhancing referrals between labour wards, Neonatal Intensive Care Units (NICUs), and outpatient departments to locate children under five years of age. Daily group education and individual counselling for mothers at discharge will be performed. Finally, the post-facility stage will focus on enhancing referral relations between facilities and the Community supported by HEWs through follow-up discharge and postnatal care cards (Table 3).
Outcome measurement
To measure the intervention’s success and population-level coverage, we will conduct a baseline household survey and an endline survey. The baseline household survey will be a representative sample survey and will answer essential questions crucial for measuring outcomes. The primary outcome of the implementation study will be to determine the proportion of children under five years of age with pneumonia who are identified and receive treatment according to the standard protocol. The secondary outcome of the implementation study will be clinical deterioration, defined as the emergence of any sign of critical illness.
For young infants aged <2 months: Critical illness (unable to feed at all, history of convulsions/convulsing at present, or no movement at all). For children 2-59 months old: Severe pneumonia or very severe disease (any danger sign, stridor in a calm child, oxygen saturation <90%, new sign of severe infection, no improvement by day 2, not recovered by day 8 for young infants aged 7–59 days with fast breathing, and not recovered by day 5 for children aged 2–59 months with chest indrawing). Additional secondary outcomes: pill count will measure the re-emergence of any presenting sign within 14 days of treatment initiation, death within 14 days of treatment initiation, treatment failure, and adherence to therapy.
Comparison
The model’s performance will be assessed against a predefined success criterion of 80% coverage of effective pneumonia treatment.
Sample size
Children 2-59 months old with chest indrawing pneumonia
In the enhanced management of pneumonia in community (EMPIC) cluster randomised controlled trial (RCT), a 4.3% treatment failure rate was reported.13 However, we assumed a 6% treatment failure rate by day 14 of enrolment in the program settings. Using the standard formula for a single proportion, a 3% margin of error, and 95% confidence level, the required sample size was 238 non-hypoxaemic children aged 2-59 months with chest indrawing pneumonia per site. Assuming 5% lost to follow-up, a total of 250 non-hypoxaemic 2-59 months children with chest indrawing pneumonia will be enrolled, given oral amoxicillin, and followed up for outcome assessment at each site.
Young infants 7-59 days old with fast-breathing pneumonia
In the EMPIC cluster RCT,14 a treatment failure rate of 5.4% was reported. However, we assumed an 8% treatment failure rate by day 14 of enrolment in the programme settings. Using the standard formula for a single proportion, a 4% margin of error, and a 95% confidence level, the required sample size is 176 non-hypoxemic young infants 7-59 days old with fast breathing pneumonia for descriptive analysis per site. Assuming 5% lost to follow-up, a total of 185 non-hypoxaemic young infants 7-59 days old with fast breathing will be enrolled, given oral amoxicillin, and followed up for outcome assessment at each site.
The sample size for cross-sectional surveys
In the EMPIC cluster RCT,14 the baseline cross-sectional survey showed that about 25% of caregivers sought treatment from appropriate healthcare providers. To estimate treatment coverage, we have calculated sample sizes at varying levels of expected coverage (Table 4).
A sample of 384 under-five children with pneumonia will allow us to estimate 80% coverage of appropriate treatment with an absolute precision of 5% and a 20% non-response rate. The number of households to be surveyed at both baseline and endline will be 614. We propose rounding to 650 households as the total sample size for the study. These households will be randomly selected from the study area using a population-proportional sampling strategy.
Data management
Qualitative data will be transcribed verbatim, translated, and analysed using the Atlas.ti software. Quantitative data on outcome measurement will be collected using electronic data collection tools (REDCap) and will be cleaned regularly. A descriptive analysis will be conducted to assess coverage, and the results will be presented using various data presentation techniques. Cross-sectional survey data will be analysed to estimate the prevalence of pneumonia and coverage of pneumonia treatment. In addition, routine evaluation data will be collected to determine the prevalence of pneumonia, the coverage rate of pneumonia treatment, the effective coverage rate, and the treatment failure rate.
DISCUSSION
Despite ongoing efforts by the WHO, pneumonia treatment coverage remains suboptimal, particularly in low-resource settings. Pneumonia continues to be a leading cause of death in children under five years of age, especially in LMICs, underscoring the urgency for more effective intervention strategies.
This implementation study will highlight the necessity for effective health system strengthening approaches to identify practical, scalable, and sustainable models for delivering childhood pneumonia management services in resource-poor settings. Through close collaboration with policymakers, the research aims to ensure that the proposed solutions are practical, context-sensitive, and adaptable to local healthcare systems. The study will explore factors such as healthcare worker training, community engagement, supply chain management, and the barriers and facilitators of integrating pneumonia treatment protocols into existing primary healthcare services. By addressing these components, the research aims to overcome current barriers to coverage, such as inconsistent treatment practices and insufficient community awareness.
This implementation research will generate valuable data and practical insights to guide the development of more tailored interventions suited to the local context, addressing barriers such as access, health worker capacity, and community engagement. The results of this study will support policymakers in shaping more effective pneumonia management programmes. They will serve as a vital resource for other LMICs facing similar healthcare challenges. The lessons learned and best practices identified could be adapted across settings, ultimately supporting global efforts to reduce morbidity and mortality from pneumonia among vulnerable populations.
This implementation research will be carried out following the two-year devastating and bloodiest war in the Tigray region, which has left nearly 90% of health facilities either entirely or partially non-functional.29 Thus, implementing this protocol will provide unique perspectives and lessons that resonate with a health system that has experienced war and is emerging into a post-war context. The findings from this study would be beneficial for scaling up this intervention in conflict-affected areas, which are increasingly common worldwide. This protocol can serve as both a guiding framework and a source of direction in situations where infrastructure is poor due to war and the need to rebuild the healthcare system. At the same time, it will address the need to provide healthcare services despite the shortage of trained healthcare providers, challenges in hiring new staff, and the ongoing need for capacity building through training in low-resource settings.
Ethical Consideration
Ethical approval was obtained from the WHO Ethics Committee under reference number WHO 2023/1358129-0, and from the Mekelle University, College of Health Sciences, Institutional Review Board (IRB) under reference number MU-IRB 1947/2023. Support letters have also been obtained from the Tigray Regional Health Bureau and District Health Offices. Individual consent from patients/families for routine health services will not be requested, as the intervention will be provided in accordance with the existing health system protocol. Written informed consent will be taken from mothers, caregivers, and health workers for the collection of qualitative and quantitative data. Data will be deidentified before analysis.
Acknowledgements
The authors would like to thank the Ministry of Health of Ethiopia and the Tigray Regional Health Bureau for their unwavering support in conducting this implementation research.
Funding
The study is funded by the Gates Foundation (#INV-008068) through a grant to the World Health Organization. The funders have no role in the study design or in the collection, analysis, or interpretation of the data. The funders do not write the report and have no role in the decision to submit the paper for publication.
Author contributions
AAM and AL are the principal investigators of the study. AAM and AL, in consultation with SAQ and YBN, conceived and designed the study. AAM, AL, SAQ, and YBN wrote the protocol. SNW, AHB, and AGH contributed to the development and write-up of the protocol. AAM, AL, LMW, and TTA adapted the protocol into a manuscript. All authors read and reviewed the first draft of the manuscript and contributed to the subsequent versions of the manuscript. All authors read and approved the final manuscript.
Disclosure of interest
The authors completed the ICMJE Disclosure of Interest Form (available upon request from the corresponding author) and disclose no relevant interests. We declare no competing interests. YBN is a staff member of the World Health Organization. The authors alone are responsible for the views expressed in this article and they do not necessarily represent the views, decisions or policies of the institutions with which they are affiliated.
Correspondence to
Name: Araya Abrha Medhanyie
Institution: School of Public Health, College of Health Sciences, Mekelle University, Mekelle, Tigray, Ethiopia
Address: Mekelle, Tigray, Ethiopia
Email: araya.medhanyie@mu.edu.et
.png)
.png)
.png)
.png)